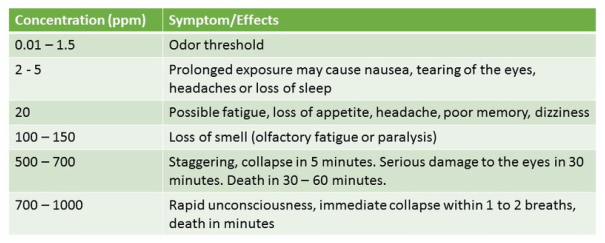
Well it all goes back to how fast bacteria reproduce. The heterotrophic (carbonaceous) or just simply BOD removing microbes in wastewater tend to grow very quickly. In making bioaugmentation products, you can have a bioreactor reach maximum population density within 24 hours with many Bacillus and Pseudomonas cultures that can double every 30 - 60 minutes depending upon substrate and environmental conditions. The fastest reproduction time is called µ(max) or maximum growth rate. For AOB, nitrifiers, growth rate is measured in hours - 7 - 8 hours in laboratory environments. In wastewater facilities you are looking at 10 - 12 hours as maximum growth rate.
Now how does this lead back to midge larvae causing more problems with effluent ammonia numbers? Simply, the midge larvae consume bacteria in the form of floc. They eat all microbes present including heterotrophic and ammonia oxidizing bacteria. Since the AOB reproduce as a slower rate, the percentage of AOB drops more than for the faster growing heterotrophic bacteria. Therefore, while we lose total MLSS (biological solids) to the larvae’s consumption, the AOB bacteria a disproportionately impacted.




 RSS Feed
RSS Feed