Most clarifiers are designed for between 2 - 4 hours hydraulic residence time, solids are recycled at a rate to prevent long solids residence times. A rule-of-thumb is to keep bed depths at 2 feet. With most clarifiers, a 2 foot bed recycles sufficiently concentrated MLSS while also preventing anaerobic conditions in the clarifier. Systems with bulking or mechanical problems often start to have trouble with 2 foot bed depths. Another problem with running deep clarifier beds is the potential for rake problems, usually torque on the motor.
Good process monitoring habits for secondary clarifiers:
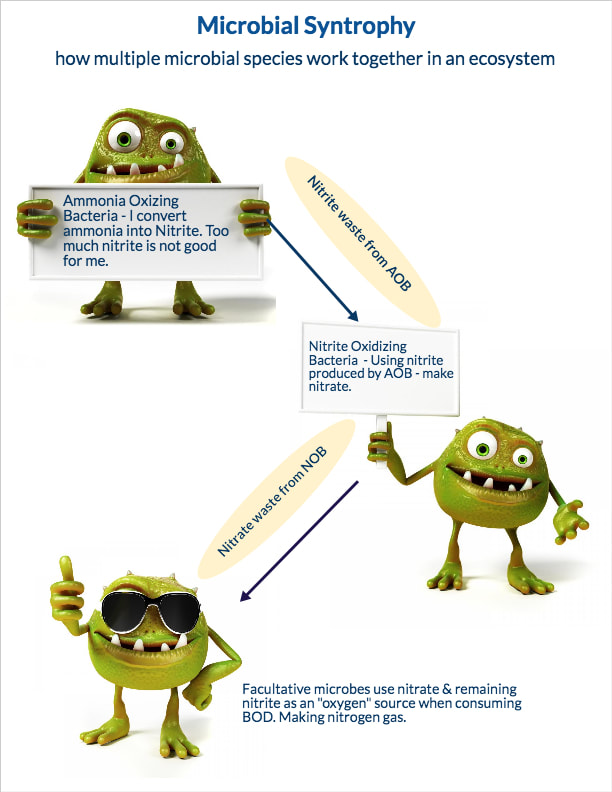
- Run SV30 and SVI on the MLSS to understand changes in settling rates. This can show potential problems in the secondary clarifiers early on. It will also inform operators on the need to add coagulants or flocculants to improve settling rates.
- Use a manual bed depth measuring tool and/or sonic levels to monitor bed depth every shift
- Return sludge concentration (RAS) concentrations should be checked
- Monitor rake torque
- Visually check for solids carryover, billowing, floating solids, scum, and problems with equipment.



 RSS Feed
RSS Feed