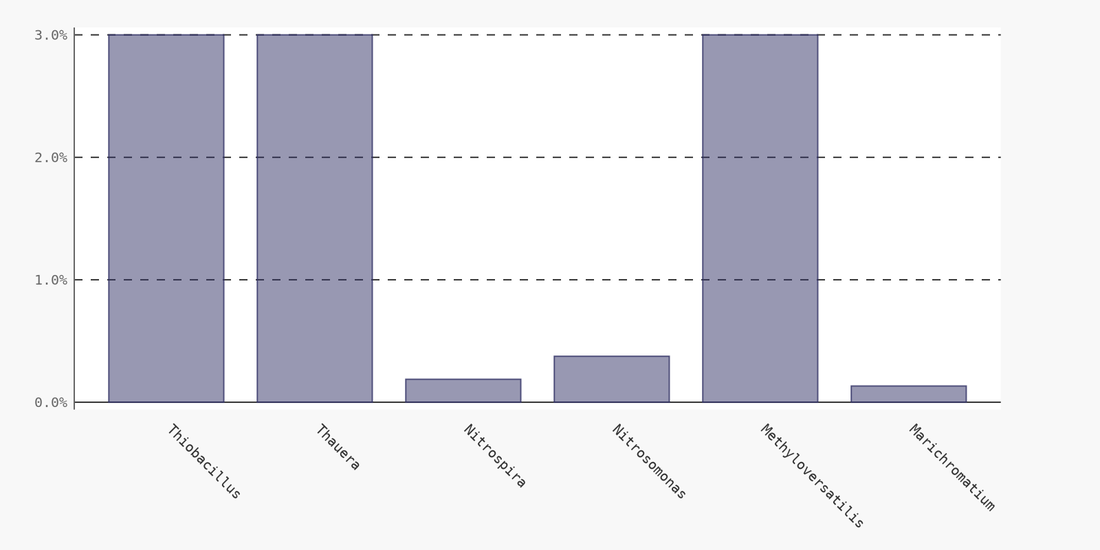
I have recently read multiple peer reviewed papers that document the ability of Nitrosomonas to utilize various organic compounds as an energy source with nitrite as an alternative electron acceptor - producing N2 gas as a final product. This ability to use their waste product from aerobic reactions as an electron acceptor reveals how Nitrosomonas can survive in systems with substantial anoxic periods.
Here is a link to a 2009 paper with more details:
https://www.ncbi.nlm.nih.gov/pubmed/19452213



 RSS Feed
RSS Feed